Vous êtes ici : ILVFRRechercheMolécules, Interactions, Matériaux (MIM)Interactions@MIM
- Partager cette page :
- Version PDF
Intéractions@MIM
L’activité du pôle « Interactions » repose sur l’utilisation de techniques de caractérisation avancées de laboratoire comme la diffraction des rayons X sur poudre ou monocristal, et la spectroscopie RMN du liquide ou du solide. Les membres du pôle mènent également des investigations structurales à partir des grandes infrastructures de recherche telles que les sources de rayonnement synchrotron (SOLEIL, ESRF, DESY) et de neutrons (ILL) ou les spectromètres RMN à très hauts champs. L’expertise du pôle couvre aujourd’hui les méthodes RMN avancées, la diffraction des RX (poudre et monocristal), la diffusion aux petits angles (SAXS/SANS) ou totale (PDF en collaboration), et plus récemment la spectroscopie d’absorption X (EXAFS, XANES en collaboration).
Spectroscopie RMN
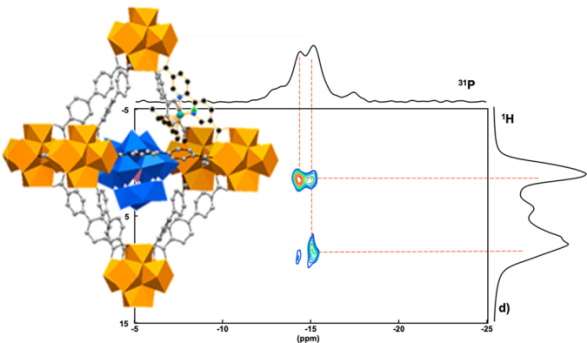
La RMN est utilisée pour la caractérisation à l’échelle locale de la structure et des phénomènes dynamiques des molécules en solution et des matériaux solides cristallisés ou amorphes. Dans ce contexte, l’équipe développe une activité de RMN solide pour des problématiques liées soit aux matériaux poreux (zéolithes, MOFs) et leurs composites (POM@MOF, nanobioMOF) soit aux matériaux moléculaires à base de POMs. L’équipe MIM est aussi reconnue pour son expertise en RMN du liquide, et plus particulièrement pour l’utilisation d’approches multinucléaires et de techniques multidimensionnelles comme la DOSY (Diffusion Ordered SpectroscopY).
Publications récentes
Diffraction des rayons X
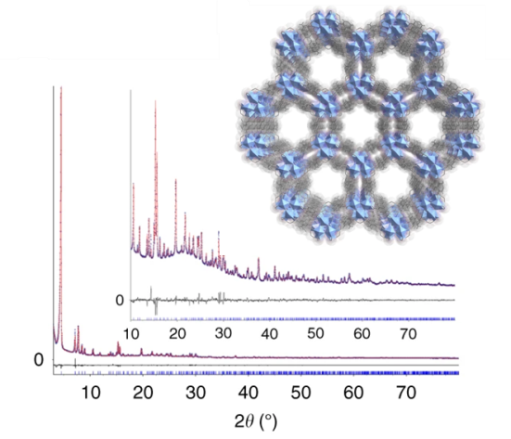 Les solides hybrides poreux étudiés dans le pôle « Matériaux » sont généralement obtenus à l’état pulvérulent. La connaissance des relations structure/propriétés étant un prérequis à leur utilisation rationnelle, la résolution structurale ab initio à partir de la diffraction des rayons X par la poudre s’avère donc indispensable. En fonction de la complexité des structures étudiées, les données issues des sources conventionnelles mais également de synchrotrons (SOLEIL et ESRF) sont utilisées. L’équipe MIM participe également au développement de méthodes visant à analyser des microcristaux par cristallographie sérielle.
Les solides hybrides poreux étudiés dans le pôle « Matériaux » sont généralement obtenus à l’état pulvérulent. La connaissance des relations structure/propriétés étant un prérequis à leur utilisation rationnelle, la résolution structurale ab initio à partir de la diffraction des rayons X par la poudre s’avère donc indispensable. En fonction de la complexité des structures étudiées, les données issues des sources conventionnelles mais également de synchrotrons (SOLEIL et ESRF) sont utilisées. L’équipe MIM participe également au développement de méthodes visant à analyser des microcristaux par cristallographie sérielle.
Publications récentes
Diffusion aux petits-angles (SAXS/SANS)
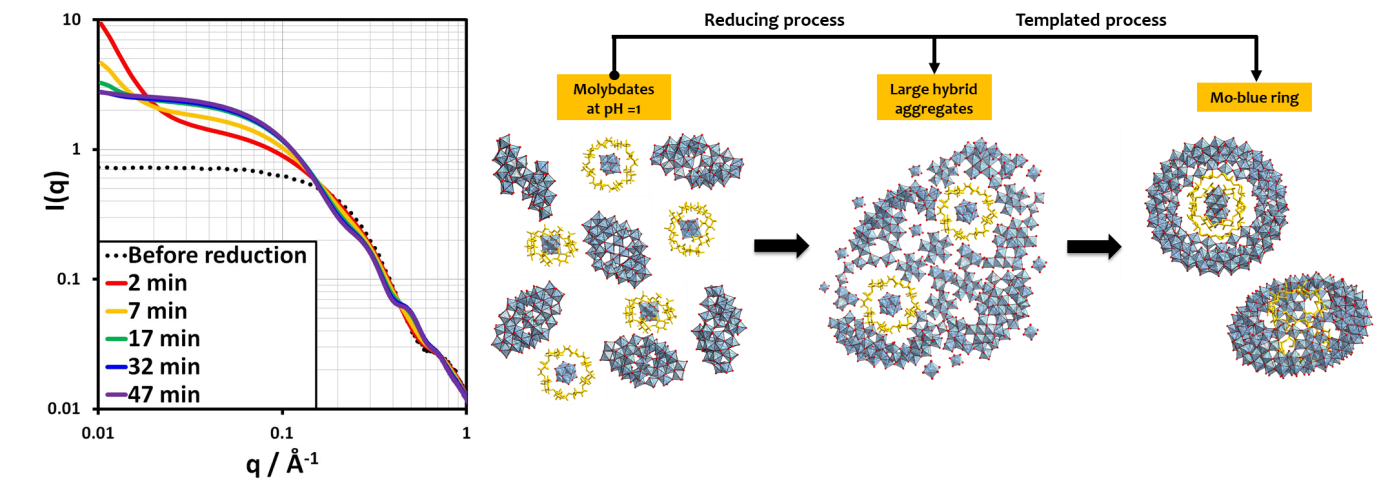 Afin de répondre aux défis associés à la caractérisation en solution d’agrégats de dimensions allant de 1 à 100 nm, l’équipe a étendu son champ d’expertise aux techniques de diffusion aux petits angles (SAXS/SANS). Ces techniques, qui apportent des informations essentielles sur les processus de croissance et d’agrégation de particules ou d’objets moléculaires en solution, ont permis à l’axe « Molécules » de développer de nouvelles thématiques de recherche centrées sur la matière molle et ses propriétés.
Afin de répondre aux défis associés à la caractérisation en solution d’agrégats de dimensions allant de 1 à 100 nm, l’équipe a étendu son champ d’expertise aux techniques de diffusion aux petits angles (SAXS/SANS). Ces techniques, qui apportent des informations essentielles sur les processus de croissance et d’agrégation de particules ou d’objets moléculaires en solution, ont permis à l’axe « Molécules » de développer de nouvelles thématiques de recherche centrées sur la matière molle et ses propriétés.






